Avance® – FDA-Approved Acellular Nerve Allograft-arwx
Repair peripheral nerves with an FDA-approved
bioactive graft that provides both the structure and
biochemical cues to support nerve regeneration
Avance is:

A biologic scaffold
Provides an extracellular roadmap and bioactive laminin to support nerve regeneration. Laminin has been shown in both in vitro assays and animal studies to be bioactive.
The exact mechanism of action is unknown.

Clinically proven
Demonstrated improvement in static two-point discrimination (s2PD) following nerve repair.*

Designed for size-matching
Available in multiple lengths and diameters to best match the native nerve diameter, provide gap length coverage and more precisely match the native nerve’s fascicular structure.
*As measured by s2PD at 12 months in RECON (N=220) which evaluated the repair of sensory nerve discontinuities ≤ 25 mm. The reported LS mean (95% CI) for Avance and Nerve Cuff were 9.1 mm (8.11, 10.04) and 9.4 mm (8.50, 10.30) respectively.
Ordering and sizing
Avance is offered in multiple diameters and lengths to best match the native nerve diameter and provide gap length coverage.
| Code | Dimensions |
|---|---|
| 111215 | 1-2 mm x 15 mm |
| 211215 | 2-3 mm x 15 mm |
| 311215 | 3-4 mm x 15 mm |
| 411215 | 4-5 mm x 15 mm |
| 111230 | 1-2 mm x 30 mm |
| 211230 | 2-3 mm x 30 mm |
| 311230 | 3-4 mm x 30 mm |
| 411230 | 4-5 mm x 30 mm |
| Code | Dimensions |
|---|---|
| 111250 | 1-2mm x 50 mm |
| 211250 | 2-3 mm x 50 mm |
| 311250 | 3-4 mm x 50 mm |
| 411250 | 4-5 mm x 50 mm |
| 111270 | 1-2 mm x 70 mm |
| 211270 | 2-3 mm x 70 mm |
| 311270 | 3-4 mm x 70 mm |
| 411270 | 4-5 mm x 70 mm |
*Dimensions (diameter x length)
Avance provides structural and biochemical cues necessary for nerve regeneration1
Avance undergoes a proprietary process to ensure an optimal structural architecture for nerve regeneration. Cellular remnants and growth-inhibiting molecules are cleared while maintaining both the structure and bioactive laminin necessary for promoting axon regeneration.
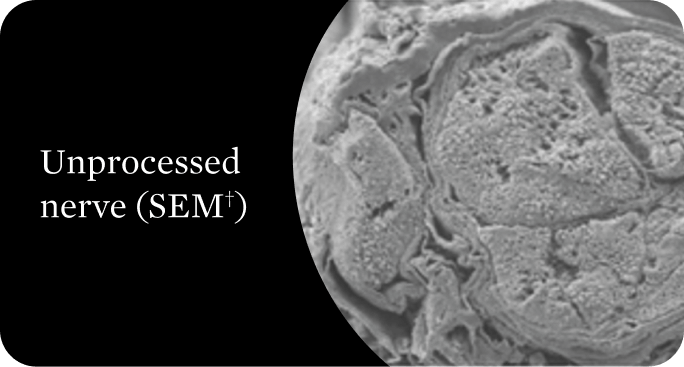
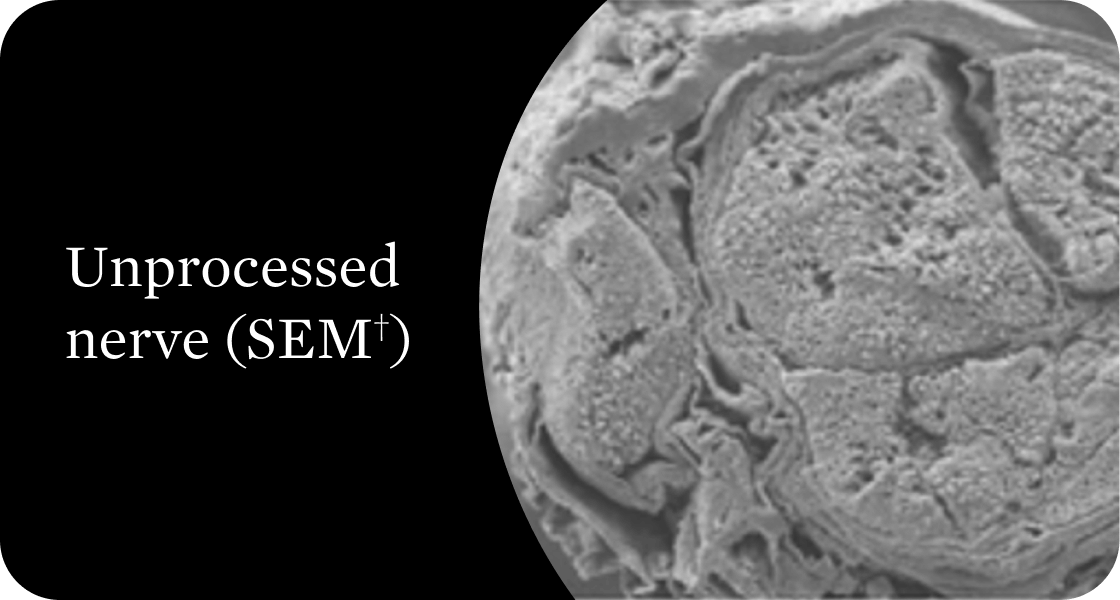
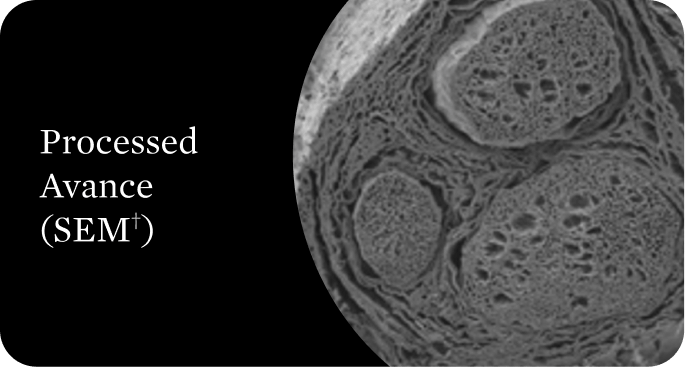
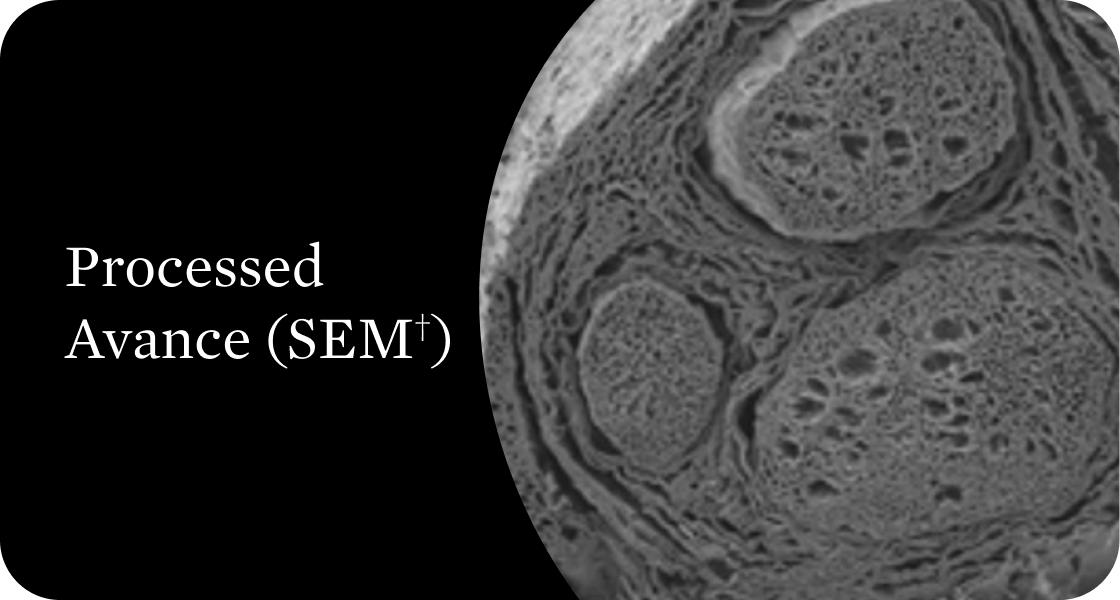
Organized structural architecture
Detergent washes clear axon remnants and other cellular debris from the endoneurial tubes while delicately preserving the entire 3D macro- and micro-architecture of the native nerve. This clears a path for Schwann cell migration and guides organized axonal regeneration.1
†Scanning electron microscope
Removal of growth inhibitors
Avance proprietary processing includes the use of an enzyme to cleave the growth-inhibiting glycosylaminoglycan (GAG) side chains of chondroitin sulfate proteoglycans (CSPGs). The GAG side chains block axon access to laminin. In animal studies, removing the chondroitin sulfate sidechains eliminated inhibitory effects, increased the amount of axon regeneration, and promoted regeneration through longer graft lengths.1,2
Why is laminin important?
Bioactive laminin is a potent promoter of neurite outgrowth.3 It plays an integral role in axon regeneration, as it contains the integrin binding sites that allow the axonal growth cone to elongate and make its way through the endoneurial tube to its distal target.3,4
Rigorous assays to verify quality
Axogen verifies the identity, purity and potency of each Avance lot. Through extensive testing and proprietary assays, we verify the quality and consistency of the nerve structure as well as its laminin bioactivity through histology and in vitro assays.
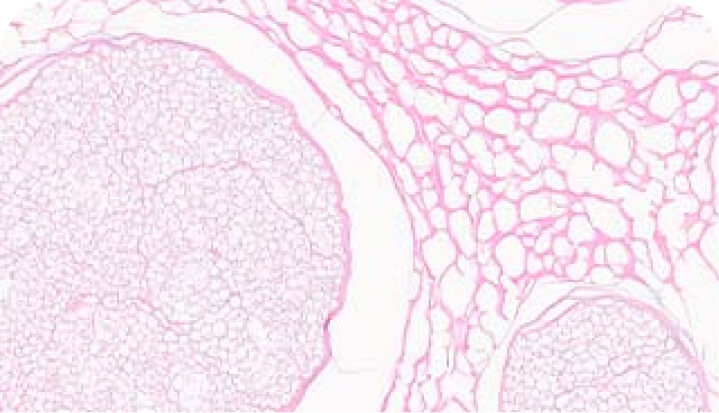
H+E stain
Verifies removal of cellular debris
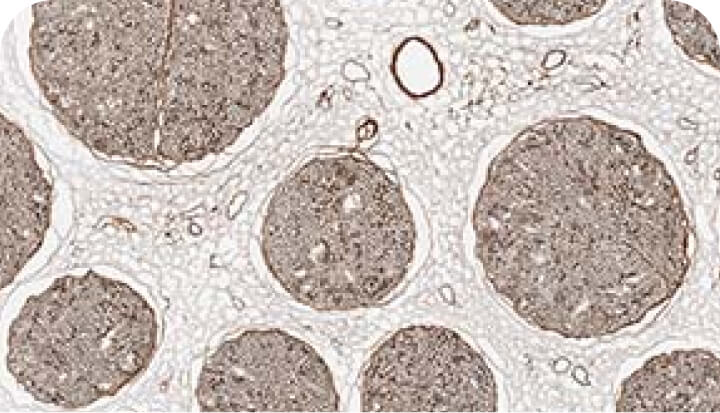
Endoneurial tube assessment
Verifies endoneurial tube quality by assessing the size and circularity of the laminin surface

Bioactive laminin assay
Verifies potency through a proprietary assay
References:
- Kasper M, et al. Bench‑to‑bedside lessons learned: commercialization of an acellular nerve graft. Adv Healthc Mater. 2020;9(16):e2000174. doi:10.1002/adhm.202000174
- Muir D. The potentiation of peripheral nerve sheaths in regeneration and repair. Exp Neurol. 2010;223(1):102-111. doi:10.1016/j.expneurol.2009.05.038
- Plantman S, et al. Integrin‑laminin interactions controlling neurite outgrowth from adult DRG neurons in vitro. Mol Cell Neurosci. 2008;39(1):50‑62. doi:10.1016/j.mcn.2008.05.015
- Danen EHJ. Integrins: An Overview of Structural and Functional Aspects. Landes Bioscience; 2013. https://www.ncbi.nlm.nih.gov/books/NBK6259/
Important Safety Information
Indications
AVANCE® is an acellular nerve scaffold indicated for the treatment of adult and pediatric patients aged one month and older with:
- Sensory nerve discontinuity (≤25 mm)
- Sensory nerve discontinuity (>25 mm); Approved under accelerated approval based on static two-point discrimination (s2PD) at 12 months in sensory nerve gaps ≤25 mm, which reasonably predicts clinical benefit. Continued approval may be contingent upon confirmatory clinical trial results.
- Mixed and motor nerve discontinuity; Approved under accelerated approval based on s2PD outcomes in sensory nerves; continued approval may be contingent upon confirmatory clinical trial results.
Warnings and Precautions
- Procedural Complications: Monitor for procedural complications, including pain, hyperesthesia, infection, implant site swelling, adhesions, hypertrophic scar formation, impaired motor or sensory function, bleeding, and neuroma formation, and manage accordingly.
- Transmission of Infectious Diseases: Because AVANCE is made from human donor tissue, it may carry a risk of transmitting infectious agents, e.g., viruses, the variant Creutzfeldt-Jakob disease (vCJD) agent, and theoretically, the Creutzfeldt-Jakob disease (CJD) agent. All infections thought to be transmitted by AVANCE should be reported to Axogen Corporation at 1-888-296-4361.
Adverse Reactions
The most common adverse reactions (≥2%) were procedural pain (4%) and hyperesthesia (3%). A serious adverse reaction (wound dehiscence) occurred in 1 patient.
